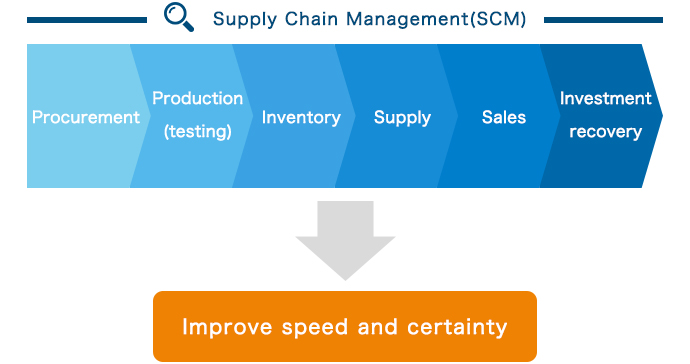
We promote Kyorin supply chain management (SCM) to raise speed and accuracy through product-by-product management (visualization) on a worldwide basis.


The Noshiro Plant
At KYORIN Pharmaceutical, we have a quality policy of maintaining stable supplies and working to reduce costs while ensuring high quality, and are currently involved in reinforcing production activities and supply chain management from a scientific standpoint. At KYORIN Pharmaceutical Group Facilities Co., Ltd. We are deepening coordination between Noshiro, Inami and Shiga plant to ensure a stable Group supply system responsive to business plans and raise the efficiency of production activities. We are working to strengthen the production system for compliance with global standards into the future. This includes further implementing Good Manufacturing Practice (GMP) quality assurance standards at all production facilities.
By working together and exceeding the bounds of the original drugs group,new drugs group and generic drugs group in addition to moving beyond the confines of Group companies, we can work on overall optimization of production within the Group.
With the goal of establishing robust earnings power that is resistant to changing conditions, we are comprehensively assessing the supply chain throughout the entire Kyorin Group and building a system for more efficient production and greater supply stability. Right now, it is essential that we promote Kyorin supply chain management (SCM) to raise speed and accuracy through product-by-product management (visualization) on a worldwide basis, from raw material procurement to production (production management and manufacturing), inventory, and supply (shipping).